Physical quantities that are sometimes
used as units alongside SI units
[this page | pdf | references | back links]
As explained in Introduction to SI units, physicists
sometimes use quantities expressed in terms of fundamental constants of nature,
alongside or instead of SI units. These fundamental constants include:
|
Kind of quantity
|
Physical quantity used as a unit measurement
|
Commonly used symbol
|
|
Speed
|
speed of light in a vacuum
|
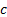
|
|
Action
|
Planck constant divided by 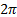 [1] [1]
|
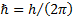
|
|
Mass
|
electron rest mass
|
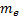
|
|
electric charge
|
elementary charge
|
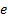
|
|
Energy
|
Hartree energy[2]
|
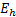
|
|
Length
|
Bohr radius[3]
|
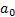
|
|
Time
|
ratio of action to energy
|
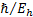
|
Notes:
[1] 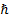 is
often also called the “reduced Planck constant”
is
often also called the “reduced Planck constant”
[2] The hartree or the Hartree energy (sometimes also
referred to by the symbol Ha) is the atomic unit of energy and is defined as 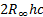 where
where 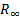 is the
Rydberg constant,
is the
Rydberg constant, 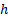 is the
Planck constant and
is the
Planck constant and 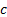 is the
speed of light in a vacuum. It is approximately the electric potential energy
of the hydrogen atom in its ground state and approximately twice its ionization
energy (the relationships are not exact because of the finite mass of the
nucleus of the hydrogen atom and relativistic corrections). It is usually used
as a unit of energy in atomic physics and computational chemistry. For
experimental measurements at the atomic scale the electron volt is more
commonly used. It also satisfies the following relationships:
is the
speed of light in a vacuum. It is approximately the electric potential energy
of the hydrogen atom in its ground state and approximately twice its ionization
energy (the relationships are not exact because of the finite mass of the
nucleus of the hydrogen atom and relativistic corrections). It is usually used
as a unit of energy in atomic physics and computational chemistry. For
experimental measurements at the atomic scale the electron volt is more
commonly used. It also satisfies the following relationships:
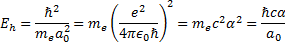
where 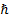 is the
reduced Planck constant,
is the
reduced Planck constant, 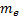 is the
electron rest mass,
is the
electron rest mass, 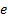 is the
elementary charge,
is the
elementary charge, 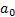 is the
Bohr radius,
is the
Bohr radius, 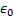 is the
electric constant (i.e. permittivity of free space),
is the
electric constant (i.e. permittivity of free space), 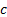 is the
speed of light in a vacuum and
is the
speed of light in a vacuum and 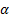 is the
fine structure constant.
is the
fine structure constant.
[3] The Bohr radius, 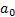 , is
approximately equal to the most probable distance between the proton and
electron in a hydrogen atom in its ground state (according to the Bohr model of
an atom). Its precise definition is (using definitions as above):
, is
approximately equal to the most probable distance between the proton and
electron in a hydrogen atom in its ground state (according to the Bohr model of
an atom). Its precise definition is (using definitions as above):
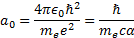
The Nematrian website makes available a range of web
functions that provide in programmatic form best estimates of the values of a
wide range of physical constants, sourced from